Latest News
INTERNATIONAL RECOGNITION PROCEDURE OFFERS HOPE FOR UK’S POSTMENOPAUSAL WOMEN.
Hypoactive Sexual Desire Dysfunction (HSDD) Perth, Australia: Lawley Pharmaceuticals, via UK-based commercial partner, Andro Pharmaceuticals Ltd, today submitted a Marketing Authorisation Application (MAA) for… Continue Reading →

Early Bird Registration: IMS World Congress on Menopause
19th World Congress on Menopause The IMS are excited to announce that registrations for the 19th World Congress on Menopause are now open! The… Continue Reading →

Podcast: Ginni Mansberg on No Filter
PODCAST: The M Word: Why So Many Women Are Angry And Weepy. Ginni is a GP in her fifties who has written a book… Continue Reading →

BMS: Menopause: Misinformation and Management
The conversation around menopause is getting louder but there is still a lot of misinformation. British Menopause Society medical professionals dispel menopause misinformation, talk… Continue Reading →

World Menopause Day – 18th October
The theme for World Menopause Day 2023 is Cardiovascular Disease World Menopause Day is held every year on the 18th October. The purpose of the… Continue Reading →
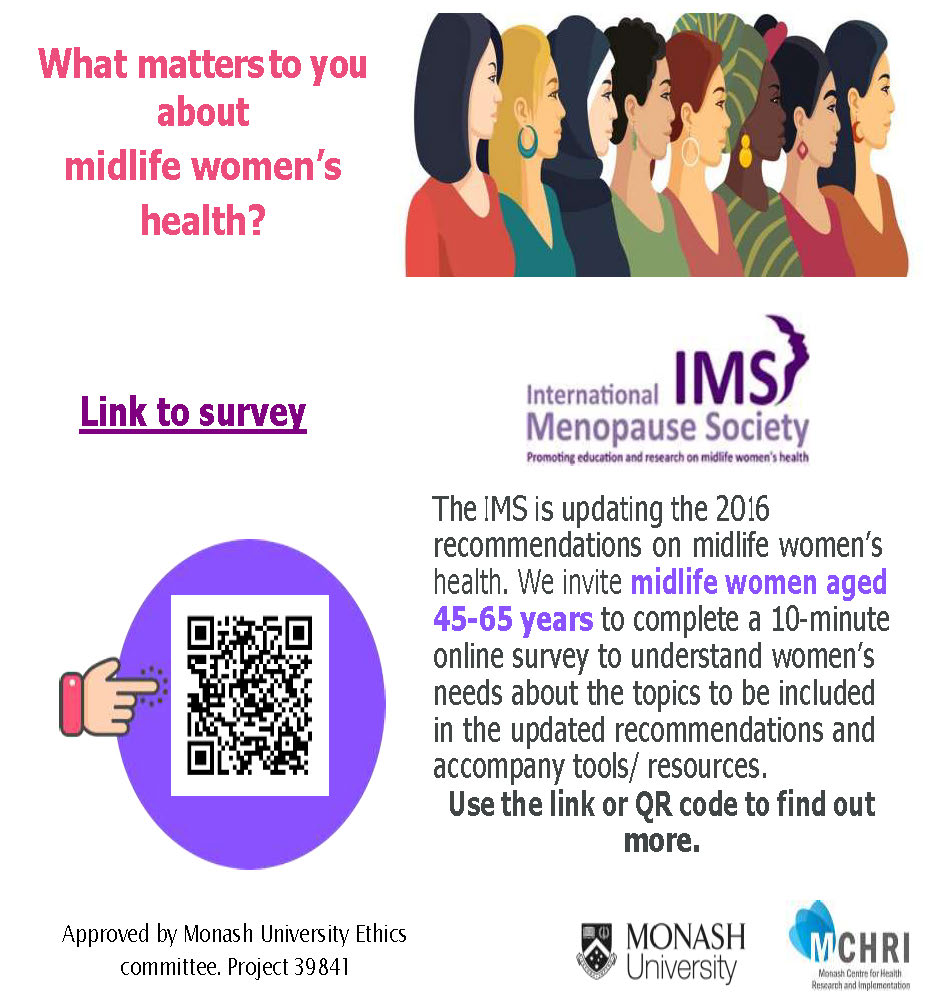

Have your say in peri-/menopause research.
VITAL is the Virtual regIstry of peri-/menopause in AusTrALia. The VITAL program aims to improve health outcomes across Australia, co-creating real-world impact via knowledge development, research,… Continue Reading →

September 7 is World Menopause & Work Day!
The theme for the second edition of this webinar is Menopause in the Workplace after COVID. EMAS launched the day in 2021 following the… Continue Reading →

Menopause Friendly Workplace Training
How Menopause Friendly Is Your Organisation? Tried, tested and trusted menopause workplace training

The 19th World Congress on Menopause will be held in Melbourne, Australia in October 2024.
Click to download the First Announcement

